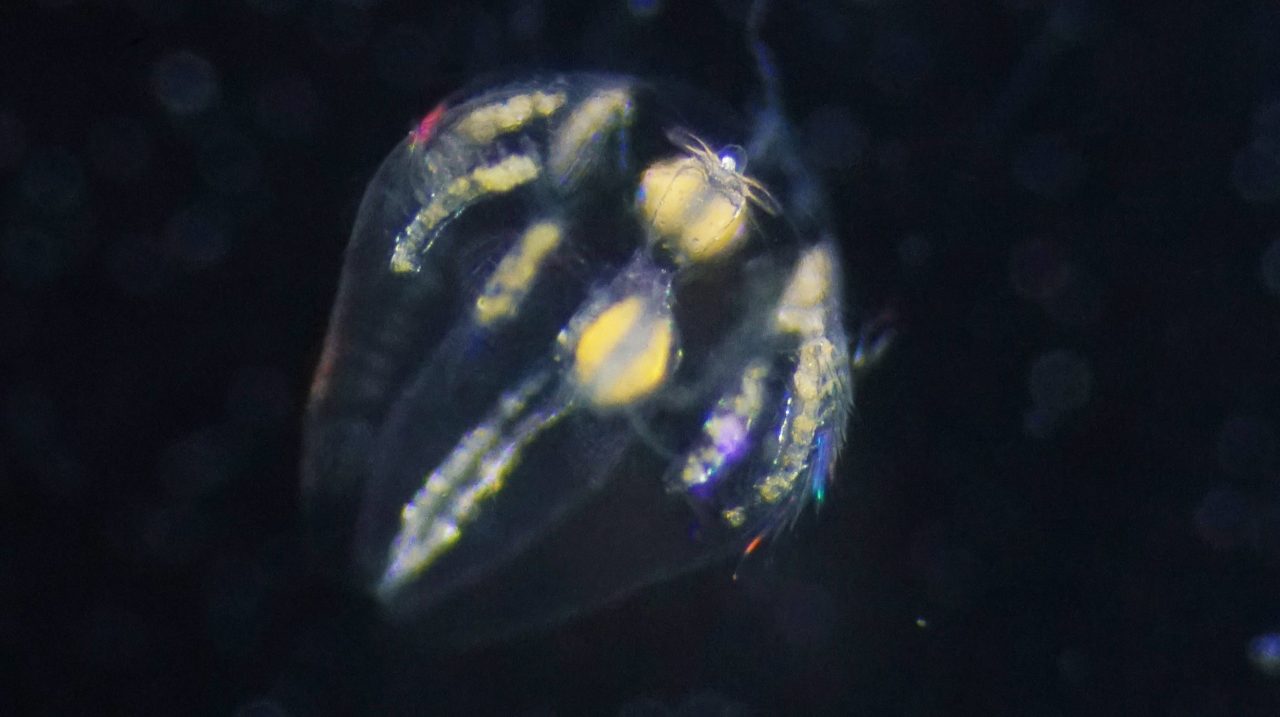
Life on Earth unfolds under gravity. To orient their bodies and navigate their environment, animals must continuously sense and respond to gravitational cues. To explain how such gravity-dependent behavioral control emerged early in animal evolution, researchers turned to ctenophores (comb jellies). These marine animals diverged from the common ancestor of animals more than 550 million years ago. Although they possess neurons, they lack a centralized nervous system, such as a brain or ganglia. Yet they can sense gravity and precisely control their ciliated comb rows, stabilizing posture and flexibly adjusting swimming direction.
A research team led by Dr. Kei Jokura (National Institute for Basic Biology (NIBB) and Exploratory Research Center on Life and Living Systems (ExCELLS)) and Prof. Gáspár Jékely (Centre for Organismal Studies (COS), Heidelberg University) examined the cellular organization and neural connectivity of the ctenophore gravity-sensing organ, the statocyst. In work published in eLife, they used volume electron microscopy to reconstruct the larval statocyst in three dimensions and generate its complete connectome. Reconstructing more than 1,000 cells across 12 distinct cell types, the team found that the entire neural network is composed of just three large, morphologically complex, multinucleated neurons.
“It was striking to find that the entire network is built from only three neurons,” says Jokura.
Previous studies had suggested neural involvement in controlling the balancer cilia, but the circuit architecture itself had remained unknown. “The statocyst consists of a statolith and four bundles of cilia known as balancer cilia,” Jokura explains. “These balancer cilia beat spontaneously, and their amplitude changes with body tilt. Mechanical signals are transmitted from the balancer cilia to the comb rows, ultimately regulating swimming behavior.” These three neurons form an interconnected network that modulates this beating of the balancer cilia.
Using high-speed imaging, the researchers then examined how this activity is controlled. Ciliary arrest events showed slight temporal delay, whereas the reactivation occurred almost simultaneously. Integrating these dynamics with the reconstructed circuit suggests that distinct neurons independently regulate the arrest and restart phases of ciliary movement. “The statocyst is not simply a relay,” Jokura adds. “It is an active, integrative circuit that dynamically coordinates ciliary activity.”
These findings provide new insight into the fundamental organization of nervous systems. Nervous systems are often viewed as centralized structures that collect sensory inputs and issue motor commands. Here, however, a decentralized nerve net achieves precise motor control without a central integrative hub. More broadly, the results suggest that neural circuits can operate as self-organized control systems, generating coordinated outputs through network-level interactions. This challenges the traditional linear view of nervous system evolution, in which simple nerve nets gradually gave rise to centralized brains. Instead, sophisticated integrative mechanisms may already have existed in early-diverging animal lineages.
The team now aims to uncover the electrophysiological and molecular basis of this network, to understand how diverse sensory inputs are integrated within the statocyst.
Paper information
Journal Name: eLife
Title of original paper: Neural Connectome of the Ctenophore Statocyst
Authors: Kei Jokura, Sanja Jasek, Lara Niederhaus, Pawel Burkhardt, Gáspár Jékely
Publication Date: February 17, 2026
DOI: https://doi.org/10.7554/eLife.108420.3

