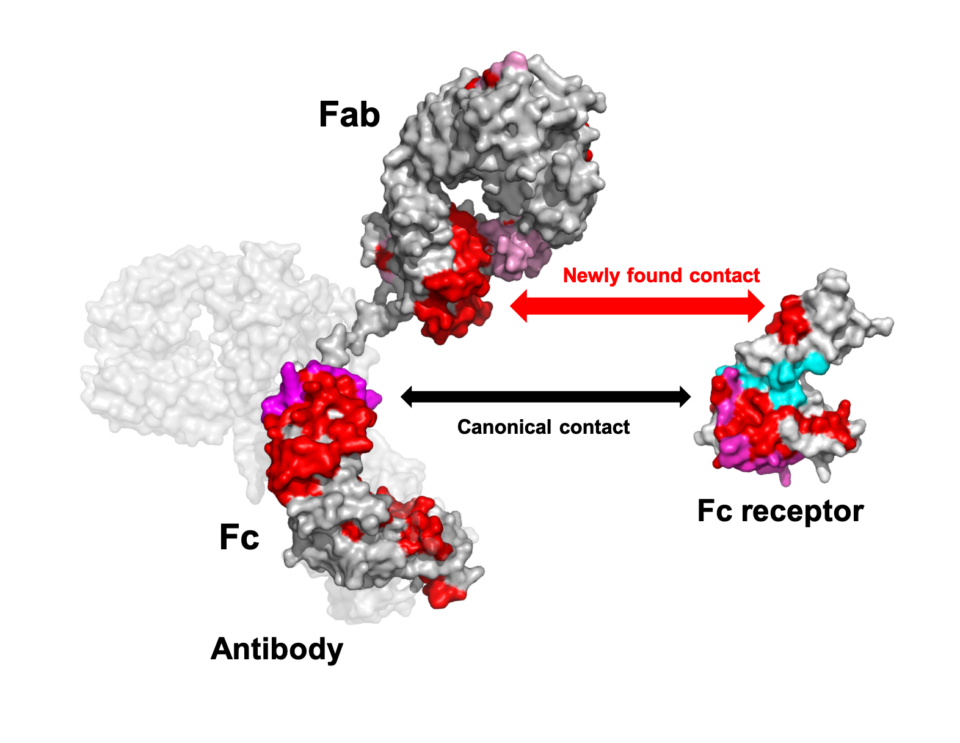
Antibody binds Fc receptor not only with its Fc trunk but also through its Fab arm
In our immune system, antibodies recognize viruses, bacteria and even cancer cells through their Fab arms, initiating recruitment of leucocytes for the destruction of these invaders. The recruitment is mediated by receptors on the leucocytes which, to date, have been supposed to bind the Fc portion of the antibody and have therefore been termed Fc receptors. This textbook view has been established based on cumulative data obtained primarily using Fab and Fc fragments cleaved from antibody molecules.
The collaborative groups including researchers at Nagoya City University, National Institutes of Natural Sciences (ExCELLS/IMS), and Osaka University reassessed this paradigm employing modern biophysical techniques. Using high-speed atomic force microscopy and hydrogen-deuterium exchange mass spectrometry, they revealed that, in addition to the canonical Fc-mediated contact, the Fab portion of the antibody is directly involved in binding to a cognate Fc receptor.
Enhancement of Fc receptor binding by molecular engineering is a promising strategy to improve the functional efficacy of therapeutic antibodies for cancer treatments. Such attempts have thus far mainly focused on the Fc trunk of the antibody. The groups’ findings will open up a new avenue for developing therapeutic antibodies with enhanced binding properties to the Fc receptor by engineering approaches targeting the previously unknown contact sites in their Fab arms.
Published online Date
August 16, 2019
Journal
Journal: Scientific Reports
Title: The Fab portion of immunoglobulin G contributes to its binding to Fcγ receptor III
Author: Rina Yogo, Yuki Yamaguchi, Hiroki Watanabe, Hirokazu Yagi, Tadashi Satoh, Mahito Nakanishi, Masayoshi Onitsuka, Takeshi Omasa, Mari Shimada, Takahiro Maruno, Tetsuo Torisu, Shio Watanabe, Daisuke Higo, Takayuki Uchihashi, Saeko Yanaka, Susumu Uchiyama & Koichi Kato
DOI: 10.1038/s41598-019-48323-w
Article URL: http://dx.doi.org/10.1038/s41598-019-48323-w
Eurekalert! URL: https://eurekalert.org/pub_releases/2019-08/ncu-ifa082119.php
Researcher URL: http://www.phar.nagoya-cu.ac.jp/hp/sbk/index_e.html
Contact
Koichi KATO
Graduate School of Pharmaceutical Sciences, Nagoya City University
3-1 Tanabe-dori, Mizuho-ku, Nagoya 467-8603, Japan
TEL:+81-52-836-3447
FAX:+81-52-836-3447
E-mail:kkato_at_phar.nagoya-cu.ac.jp
Please replace the “_at_” with @

