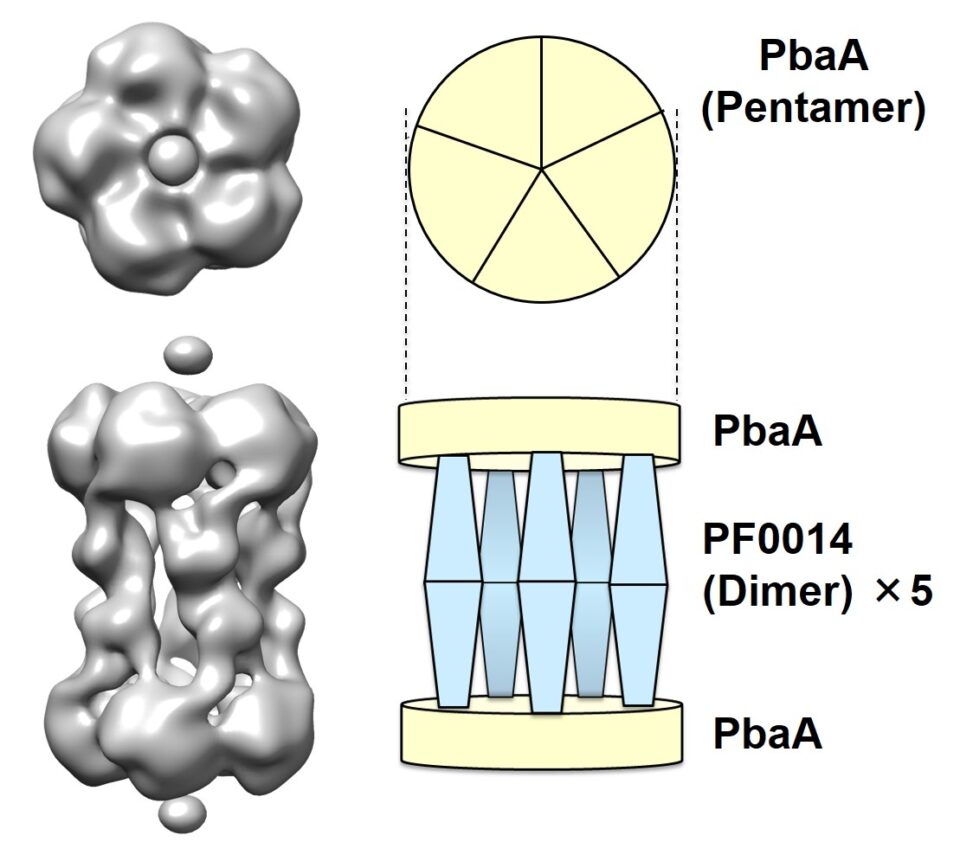
A unique archaeal protein complex having a spacious center surrounded by five columns
Proteins often form assemblies and thereby perform sophisticated functions in cells as best exemplified by proteasomes, which are huge enzyme complexes functioning as proteolytic machines. In eukaryotes, this proteasome formation is not a spontaneous process but is assisted by several other proteins, termed proteasome-assembling chaperones. Paradoxically, archaeal genomes encode proteasome-assembling chaperone homologs, denoting a shared ancestry between genes, although archaeal proteasome formation is a spontaneous process not requiring these chaperones. Therefore, the functional roles of the archaeal chaperone-like proteins remain unknown. The collaborative groups, including researchers at Exploratory Research Center on Life and Living Systems (ExCELLS), Institute for Molecular Science (IMS), and National Institute for Physiological Sciences (NIPS) of National Institutes of Natural Sciences found that a chaperone-like protein originating from a hyperthermophilic archaeon together with another protein from the same species, whose function is also unknown, are assembled together into unique structures. The integrated biophysical data they obtained using native mass spectrometry, solution scattering, high-speed atomic force microscopy, and electron microscopy, along with atomic structure modeling, revealed that this complex forms a five-column tholos-like architecture, harboring a large central cavity, which can potentially accommodate biomolecules, such as proteins. This characteristic architecture of archaeal protein complex provides insight into the molecular evolution between archaeal and eukaryotic proteins. Furthermore, their findings offer a novel framework for designing functional protein cages as molecular warehouses or shelters that are stable in high temperatures.
Journal
Journal: Scientific Reports
Title: Supramolecular tholos-like architecture constituted by archaeal proteins without functional annotation
Author: Maho Yagi-Utsumi, Arunima Sikdar, Chihong Song, Jimin Park, Rintaro Inoue, Hiroki Watanabe, Raymond N. Burton-Smith, Toshiya Kozai, Tatsuya Suzuki, Atsuji Kodama, Kentaro Ishii, Hirokazu Yagi, Tadashi Satoh, Susumu Uchiyama, Takayuki Uchihashi, Keehyoung Joo, Jooyoung Lee, Masaaki Sugiyama, Kazuyoshi Murata, and Koichi Kato.
DOI: 10.1038/s41598-020-58371-2
Eurekalert! URL: https://eurekalert.org/pub_releases/2020-02/nion-agt020320.php
Contact
Koichi Kato
Exploratory Research Center on Life and Living Systems (ExCELLS)/
Institute for Molecular Science, Natural Institutes of Natural Sciences
TEL: +81-564-59-5225
E-mail: kkato_at_excells.orion.ac.jp (Please replace the “_at_” with @)

